Fluorescence is a luminescence that is mostly found as an optical phenomenon in cold bodies, in which the molecular absorption of a photon triggers the emission of another photon with a longer wavelength. The energy difference between the absorbed and emitted photons ends up as molecular vibrations or heat.
Fluorescence is a luminescence that is mostly found as an optical phenomenon in cold bodies, in which the molecular absorption of a photon triggers the emission of another photon with a longer wavelength. The energy difference between the absorbed and emitted photons ends up as molecular vibrations or heat.
Usually the absorbed photon is in the ultraviolet range, and the emitted light is in the visible range, but this depends on the absorbance curve and Stokes shift of the particular fluorophore. Fluorescence was coined by George Gabriel Stokes in his 1852 paper, the name was given as a description of the essence of the mineral fluorite, composed of calcium fluoride, which gave a visible emission when illuminated with "invisible radiation" UV radiation.
Cystoscopy
Endoscopy of the urinary bladder via the urethra is called cystoscopy. Diagnostic cystoscopy is usually carried out with local anaesthesia. General anaesthesia is sometimes used for operative cystoscopic procedures. When a patient has a urinary problem, the doctor may use a cystoscope to see the inside of the bladder and urethra.
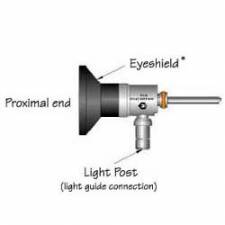
The urethra is the tube that carries urine from the bladder to the outside of the body. The cystoscope has lenses like a telescope or microscope. These lenses let the doctor focus on the inner surfaces of the urinary tract. Some cystoscopes use optical fibres (flexible glass fibres) that carry an image from the tip of the instrument to a viewing piece at the other end. The cystoscope is as thick as a pencil and has a light at the tip. Many cystoscopes have extra tubes to guide other instruments for surgical procedures to treat urinary problems.
There are two main types of cystoscopy - flexible and rigid - differing in the flexibility of the cystoscope. Flexible cystoscopy is carried out using local anaesthesia on both sexes. Typically, lidocaine gel (such as the brand name Instillagel) is used as an anaesthetic, instilled in the urethra. Rigid cystoscopy can be performed under the same conditions, but is generally carried out under general anaesthesia, particularly in male subjects, due to the pain caused by the probe.
A doctor may recommend cystoscopy for any of the following conditions:
- Interstitial Cystitis
- Frequent urinary tract infections
- Blood in the urine (hematuria)
- Loss of bladder control (incontinence) or overactive bladder
- Unusual cells found in urine sample
- Need for a bladder catheter
- Painful urination, chronic pelvic pain, or interstitial cystitis
- Urinary blockage such as from prostate enlargement, stricture, or narrowing of the urinary tract
- Stone in the urinary tract
- Unusual growth, polyp, tumour, or cancer
Male and female urinary tracts.
 The top two images show the interior of the bladder of a male patient.
The top two images show the interior of the bladder of a male patient.
In the top-right image, the cystoscope has been bent within the bladder to look back on itself. The bottom two images show an inflamed urethra. If a patient has a stone lodged higher in the urinary tract, the doctor may use a much finer calibre scope called a ureteroscope through the bladder and up into the urethra. (The urethra is the tube that carries urine from the kidney to the bladder). The doctor can then see the stone and remove it with a small basket at the end of a wire which is inserted through an extra tube in the ureteroscope.
For larger stones, the doctor may also use the extra tube in the ureteroscope to extend a flexible fibre that carries a laser beam to break the stone into smaller pieces that can then pass out of the body in the urine.
The doctor will gently insert the tip of the cystoscope into the urethra and slowly glide it up into the bladder. The procedure is more painful for men than for women due to the length of the urethra. Relaxing the pelvic muscles helps make this part of the test easier. A sterile liquid (water, saline, or glycine solution) will flow through the cystoscope to slowly fill the bladder and stretch it so that the doctor has a better view of the bladder wall. As the bladder reaches capacity, patients typically feel some mild discomfort and the urge to urinate. The time from insertion of the cystoscope to removal may be only a few minutes, or it may be longer if the doctor finds a stone and decides to remove it.
Taking a biopsy (a small tissue sample for examination under a microscope) will also make the procedure last longer. In most cases, the entire examination, including preparation, will take about 15 to 20 minutes. After the test, patients often have some burning feeling when they urinate and often see small amounts of blood in their urine. Occasionally, patients may feel some lower abdominal pains, reflecting bladder muscle spasms, but these are not common. (These problems should not last more than 24 hours.
Fluorescence cystoscopy
When performing at fluorescence cystoscopy, the existing endoscopic instruments must be properly complimented. The key instruments are a powerful light source for fluorescence excitation and special endoscopes (see below).

A special camera is also needed for video guided cystoscopy. All devices developed for fluorescence cystoscopy may also be used for white light cystoscopy. They can also be combined as desired with standard accessory instruments. In order to ensure both the quality and the success of fluorescence diagnosis, all technical components must be in perfect condition.
Light source and cystoscope.
Fluorescence diagnosis requires a light source that is very powerful, especially in the blue spectral range, where the PPIX (Protoporphoryn IX: molecule that accumulates in tumours) converts the excitation light efficiently into red fluorescence light. The fluorescence can be observed, only if a large portion of the excitation light is blocked by an observation filter. This filter is integrated in the endoscope.
The combination of the spectral transmission characteristics of the excitation filter, the light source, and the observation filter in the endoscope, determines the intensity of the back scattered blue excitation light seen by the observer. In order to provide an optimum contrast between fluorescent and non-fluorescent tissue, the back scattered excitation light and fluorescence light should have a compatible intensity. When combining light source and cystoscope, it is necessary to ensure that the manufacturer has matched these devices to each other. Only endoscopes with integrated observation filters can be used. Light sources and cystoscopes made by different manufacturers must never be combined with each other. The excitation filter in the light sources mounted filter will sell a simple and rapid change between standard and fluorescence cystoscopy is possible.
A foot switch is used during a fluorescence cystoscopy to switch between white light and violet blue fluorescence excitation light. If the fluorescence capable video camera is used, the footswitch is replaced by an electronic coupling between the camera and light source. This allows for switching between the two operating modes, by push buttons on the camera head.
By filtering out part of the blue spectral range, a slightly yellowish tint is seen through the endoscope. This does not, however, affect colour reproduction during a white light cystoscopy. To make sure that the excitation light is efficiently transmitted to the target tissue. It is mandatory that specific custom manufactured fluid light cables of small diameter be used, as well as optimised light transmission bundles integrated in the fluorescence capable cystoscopes. (Fluid light cables are sensitive to mechanical impact or thermal effects and must not be sterilised and autoclave). The advantages of the fluid light system include blocking of residual infrared light and lower intrinsic fluorescence. It may, however, more easily occur and components of the fluid system and of standard equipment accidentally mistaken (with each other). Also, the fluid light cable cannot be autoclaved. Fluid light cable and telescope must be stored together: if a non-fluorescence capable optical light cable is used, substantial light loss occurs. If a non-fluorescence capable telescope is used, it will not be possible to visualise porphyrin fluorescence, because of the missing observation filter, and also because the coupling between the light cable and telescope is not optimised.
The endoscopes direction of view (0, 12°, 30°, or 70°) is selected dependent on the localisation observed. It should be considered, however, the endoscopes of the larger direction of view, transferred less excitation light than those with the smaller one. Since the type of xenon short arc lamp used is subject to ageing, maintenance should be performed as soon as any reduction in light power is perceived. The life of the xenon short arc lamp is approximately 500 operating hours. Replacement of lamps is recommended after about 300 hours. The reason for this is that the light power available distally on the cystoscope not only decreases in proportion with the lamp power but also due to an increased focal spot as a result of the ageing process. As a result, the light power radiated by the lamp can no longer be optimally coupled into the fibre optic light bundle of the cystoscope.
Practical aspects of fluorescence diagnosis
Light intensity: Even if the technical equipment is in perfect condition and the application has been performed according to the rules, the image brightness in the fluorescence method still may be unsatisfactory to the eye of the operator.Possible causes:
- observation distance too long.
- Blood in irrigation fluid.
- Photo bleaching of fluorescence
- the fluorescence of urine
Distance: the brightness of the endoscopic image depends on the illumination intensity and the imaging geometry. The illumination intensity decreases with the square of the distance from the distal end of the endoscope to the tissue. This is also true of the amount of light surface unit that is collected from the tissue by the endoscope.
However, the area observed through the endoscope also increases with the square of the distance. Overall light intensity observed through the eyepiece shows a square dependence on distance. This relationship is valid from observation distance greater than about 10 mm, both for white light and fluorescent light.
The square dependence on distance means that a doubling of the observation distance causes a reduction of the image brightness of the entire viewing area to one quarter of the original brightness. With regard to the detect-ability of a tumour that covers only part of the viewing area. It must be considered that it is imaged to an area that is also decreased by one quarter. This means that to double the observation distance means only the 16th parts of the fluorescence light can be detected.
Because of the distinct wide angle visualisation of endoscopes, an absolute estimate of the distance to the bladder wall is difficult. In case of a bladder that is very large and/or overextended because of high filling pressure, the surgeon will tend to use larger distances. Under white light, the dynamics of eye or camera will be sufficient for compensating the related loss of image brightness. But the image may become too dark for a fluorescence observation. By tolerating a reduced image frame, a satisfactory image brightness can be obtained by reducing the observation distance. If, in the presence of a clear irrigation fluid, the eye is unable to see a good picture at the distance of an extended resection loop (normal bladder wall: blue to blue -- green, vascular structure is clearly visible as a dark network), this indicates a technical error in the illumination path. If the image brightness is insufficient only on the monitor, this indicates a technical error or an operating error related to the camera. Fluorescence cameras equipped with zoom lenses permit a variably adjustable image magnification. The higher the magnification is set, the darker the picture will become on the monitor. It must be emphasised, however that a necessarily short observation distance settings should not be used since this may lead to rapid photo bleaching of the site.
Results
Two consecutive studies since 1992 and evaluated a total of 247 bladder tumour patients with regard to significance and feasibility of the procedure. Sensitivity and specificity of urothelial tumour detection with white light was compared to that of fluorescent light. In the process, a biopsy related analysis of the data was performed, taking into account endoscopic findings under the white light, fluorescence findings, and histology. It was a very high correlation between biopsies taken from areas with either positive fluorescence or negative fluorescence and histological findings. From this correlation, a sensitivity of more than 95% and a specificity of 68.5% was calculated.
The rate of residual tumour could be reduced significantly by nearly 40% using fluorescence. Moreover, statistical benefit could be shown for low-risk patients and patients with tumours.
Summary
The use of fluorescence endoscopy for bladder cancer has been found to be successful in routine clinical applications. Compared to conventional endoscopy, fluorescence endoscopy permits rapid, highly sensitive detection of urothelial neoplasias, with almost no side-effects. Because of its high sensitivity, no random biopsy is necessary if the examination result was completely fluorescence negative. Fluorescence endoscopy is helpful to verify suspicious, cytological bladder washing findings. Initial results demonstrate the early detection of urothelial neoplasias.
Glossary
CCD camera: charge coupled device, the standard image sense of the video and endoscopic cameras. This term designates the microchip onto which the light falls and causes free electrons to be generated. The electrons are then electronically separated and converted to form the image signal. Fluorescence cystoscopy requires special CCD endoscopic cameras.
Colour contrast: in fluorescence cystoscopy, abnormal tissues are discriminated from the normal bladder wall by visualising a colour contrast. The red fluorescence in tumours contrasts in colour from the blue excitation light.
Excitation filter: an optical filter system in the excitation light system, high transmission in the violent part of the spectrum and blockage of the remaining, visible part of the spectrum ensures optimum fluorescence observation.
Fluid light cable: light cable to transmit in white light of fluorescence excitation light from the source to the endoscope, characterised by good transmission in the short waveband of the spectrum, and by low infrared transmission. Special small diameter fluid light cables were developed to be used for fluorescence diagnosis. Only these types of fluid light cables are compatible with the special fluorescence telescopes used in fluorescence cystoscopy.
PDD: photodynamic diagnosis, a variation of the common acronym PDT (photodynamic therapy) for the fluorescence diagnosis use of photo toxics substances (photosensitiser's) with tumours selective accumulation.
Article compiled by John Sandham IEng MIET MIHEEM
Sources:
http://en.wikipedia.org/wiki/Fluorescence_microscope
http://www.nlm.nih.gov/medlineplus/ency/imagepages/1089.htm
http://en.wikipedia.org/wiki/Fluorescent
http://kidney.niddk.nih.gov/kudiseases/pubs/cystoscopy/
http://www.doctorslounge.com/urology/procedures/cystoscopy.htm