Healthcare technology procurement, use, maintenance, and management policy is a serious issue recognised by research undertaken worldwide. The World Health Organisation carried out a global survey in 2015 on Health technology assessment. One of the findings in the report concluded that medical devices were considered almost always essential to the safety and clinical effectiveness of the healthcare system as illustrated below. 1


Some of the critical literature discussing ‘healthcare technology issues’ come from the National Audit Office, the National Patient Safety Agency, the Medicines and Healthcare Products Regulatory Agency and the World Health Organisation.
The 2015 MHRA document ‘Managing Medical Devices: Guidance for healthcare and social services organisations’ is an excellent guide for healthcare managers and describes a systematic approach to the acquisition, deployment, maintenance (preventive maintenance and performance assurance), repair and disposal of medical devices.2
Healthcare technology management is an extremely complex area within healthcare, covering the procurement, training, maintenance and governance of medical devices ranging from magnetic resonance imagers (MRI) to a simple thermometers.
Professional stakeholders (whether from procurement department buying devices, from clinical departments using devices, from maintenance departments repairing and maintaining the devices, or the governance department ensuring compliance with internal policies and external regulations) must interact to ensure they select and use devices to deliver quality health services.
Medical devices are used in Hospitals for diagnostic and therapeutic purposes. It is important that these devices are used correctly to ensure the safety of the patient; indeed, there is evidence that patients are sometimes harmed or die because devices are not used correctly. Even. Discussing the reasons for these incidents with the clinicians and nurses involved leads me to the conclusion that there is too wide a variety of devices within healthcare organisations that do the same job.
This variety makes it difficult to ensure user training is carried out on all devices. Therefore, many hospitals take the decision to only to train on the highest risk devices that can harm or kill (in the event of misuse), and allow self-certification on all other devices. This risk-based approach is the only one that is considered feasible in the short term.
It is easier to achieve standardisation if equipment is planned. Standardisation helps you to improve quality by:
- Ensuring that only products which meet the defined standards are acquired, without the need for additional cumbersome tests and checks.
Procurement and logistics:
- Limiting the number of types of spare parts, accessories, and consumables that you have to keep in stock for different types of equipment.
- Helping you to rationalise your sources of supply and supply routes, and make savings through bulk purchasing.
Skill development and performance:
- Enabling staff to become more knowledgeable about the operation and maintenance of the products, as it limits the variety of items they have to be familiar with.
After sales support:
- Giving suppliers greater incentives to provide after-sales services and establish long-term relationships with you due to the bulk sales.
Consequently, the only way to improve training, and thereby reduce the risk of harm to patients, is to standardise devices to a single equipment type from a single supplier for each type of clinical technology application. This is difficult to achieve at the moment because purchasing mechanisms, although moving towards standardisation of devices, healthcare organisations still have a long way to go.
In the guide, ‘How to Organize a System of Healthcare Technology Management’ (HTM) it describes the healthcare technology management cycles (as shown below).3
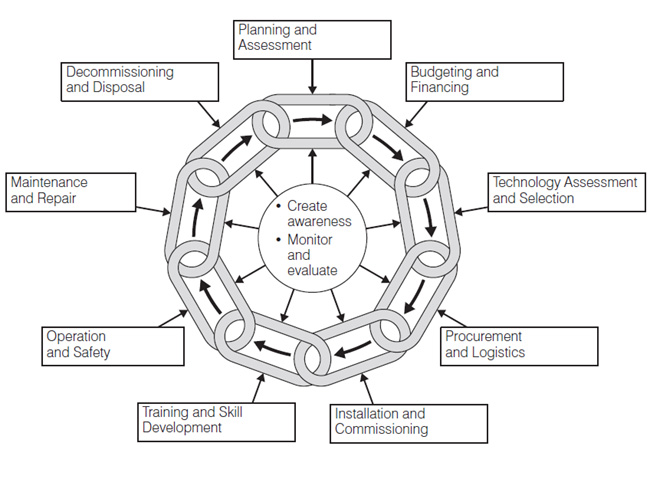
The HTM process illustrated is complex and difficult to achieve because hospitals have hundreds of different technology groups in use for different diagnostic and therapeutic purposes. It therefore makes it extremely difficult to find agreement from users within each technology group, on what devices to buy and when to buy them. There can be hundreds of device users across multiple wards and departments who all want to have a say in what they use on patients.
The age profile of medical devices can be wide, from the newest to the oldest devices across individual technology groups, which leads to some devices being less than a year old whilst other devices in the Hospital from same technology group (with the same clinical function) are more than ten years old and already past their useful life. This issue happens as a result of multiple budgets, multiple purchases by different budget holders, and limited or unplanned funding. As long as there are multiple budgets and device users continue to have individual choice, the risks will remain.
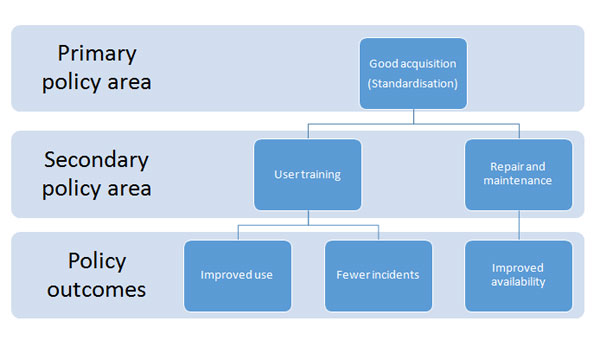
Procurement and EBME managers must work together to understand primary and secondary policy areas, and deliver policy outcomes that improve use and availability, thereby leading to fewer incidents. (as illustrated below)
Not only does this impact on patient safety, it also affects cost because the procurement manager cannot access the same level of discount when devices are bought individually.
Device users such as nurses and doctors need equipment that they are able to use safely, is up-to-date, and functions well. EBME managers, engineers and technicians also want standardisation, and want users to be trained because, with increased standardisation, they could carry less spare parts and become more expert in repairing devices, thereby reducing down-time. Moreover, with trained users there would be fewer callouts to equipment that is not at fault. EBME engineers often discuss the number of calls they receive because users do not know how to use the equipment correctly and often ‘think it is faulty’ rather than there being an actual fault.
Governance managers are also concerned about meeting inspection standards from the regulators. To achieve this, they work with internal stakeholders to ensure that healthcare technology management policy is inclusive not only of the internal requirements of the hospital (such and procurement conduct, user training and maintenance), but must also meet external regulatory requirements from the health and social care act.
In summary, it is the responsibility of a wide range of healthcare professionals to bring together their joint knowledge to innovate and improve healthcare practice.
Dr John Sandham
Bibliography
- World Health Organisation.
2015 Global Survey on Health Technology Assessment by National Authorities. [Online] 2017. - Medicines & Healthcare products Regulatory Agency.
Managing Medical Devices - Guidance for healthcare and social service organisations. London : MHRA, 2015. - Lenel, Andreas, et al.
How to organise a system of Healthcare Technology Management. London : Lewes, 2005.