Authors:
Mr. David Cook - Managing Director, Accordia PPP.
Dr. John Sandham - Chairman of ebme.co.uk & CEO TBS GB.
 It is internationally accepted that technology can be an accelerant for delivering transformative change. This can only happen if the investment in technology is planned, the staff using the technology have appropriate support and training, and the technology strategy actually delivers more productive healthcare at a lower cost.
It is internationally accepted that technology can be an accelerant for delivering transformative change. This can only happen if the investment in technology is planned, the staff using the technology have appropriate support and training, and the technology strategy actually delivers more productive healthcare at a lower cost.
The Nuffield Trust recently stated that technology can transform the delivery of healthcare if organisations not only understand the opportunity to transform services, but are also able to ‘grasp’ those opportunities and actually deliver the changes. ‘Clinically led improvement, enabled by new technology, is transforming the delivery of health care and our management of population health. Yet strategic decisions about clinical transformation and the associated investment in information and digital technology can all too often be a footnote to NHS board discussions. This needs to change. This report sets out the possibilities to transform health care offered by digital technologies, with important insight about how to grasp those possibilities and benefits from those furthest on in their digital journey’. (1)
NHS Budget reduced by £1.1Bn for capital investment
In a recent news story, the shadow health secretary said that 'Five years of Tory neglect has left many hospitals with ageing equipment and a growing bill for urgent maintenance.’ The capital budget of the NHS is used to fund repairs and replace out-of-date or broken equipment. The NHS was expected to be allocated £4.8bn to cover this area, but the Budget revealed the health service will only be receiving £3.7bn of capital budget.’ (2)
There are many innovators across healthcare organisations that can see the potential of technology to deliver change, but are still unable to deliver for reasons of lack of capital, and often, lack of people in leadership positions with the knowledge, skills and authority to champion the case for new technology and deliver the changes required.
The technology already exists to transform healthcare. What we really need are the right people able to deliver, and entrepreneurial partners willing to invest their knowledge and finances to resource delivery of the transformation as part of a business collaboration. Healthcare technology businesses are willing to work with public and private hospitals to introduce healthcare technology (new medical devices and software) that can improve patient care, staff productivity, and cost per patient.
One area in which technology solutions should deliver efficiency is in the technological integration from the home to the ward for step up and from the ward to the home for step down care models. This is only possible if the healthcare organisation recognise the need to call upon technology experts when it comes to connecting different technologies. There is a lot of evidence about how information technology can transform healthcare, but unfortunately there are also many cases where IT has failed to deliver the expected benefits. Real transformation only happens if the top management in health trusts and health boards understand the significance of involving technology experts in the decision making. Technology is developing at such a pace that non-experts may be unaware of not only technological requirements, but opportunities that can arise from harnessing the latest technological developments. This map of stakeholder relationships within the healthcare organisation is changing and the leadership required to utilise technology requires a multi-disciplinary team that includes clinical engineers and IT system engineers, in addition to the more established stakeholders from procurement, finance, and clinical backgrounds.
A Paperless NHS?
Recent reports require NHS organisations to become paperless by 2020, with “key digital information systems in place, fully integrated and utilised by October 2018”. Systems such as EPMA (Electronic Prescribing Medicines Administration) and Radio-Frequency ID tracking are just some of the systems cited in these reports. (3) (4)
Healthcare organisations have 100’s of different types of medical technology (medical equipment and medical software), and many different types of information technology systems. There are real benefits that can come from linking the medical equipment to the IT system and allowing the patient data to be ‘pushed’ into the patient record. For example, EPMA systems can ensure that the prescribed medication for each patient is allocated to the right patient record. Modern infusion delivery systems can facilitate “drug library” software in which all concentrations of drugs are held in the memory of the pump, and by linking the pump to the patient via a barcode, the patient prescription can be uploaded directly to the pump. Time of entering data and checking is reduced, with the removal of the risk of human transcription errors.
Furthermore, the actual medication delivered is automatically uploaded into the Electronic Patient Record. Such systems were not practicable prior to networking of devices; large hospitals may have 1000s of infusion pumps, and drug libraries will require regular updating so the only way to manage this is by sending the data over the hospital network so that all devices can be harmonised to the same configurations.
Whilst the technology is available now to do this, there is sometimes a misconception that it can happen simply by implementing a large IT system. Healthcare institutions need to make strategic decisions about which devices they wish to connect in to the hospital network, and plan investment so that in addition to the IT network, the 1000s of medical devices are specified so as to work with the network. Wifi connectivity does not come as standard on most medical devices, so strategic decisions are needed to ensure that once we have our EPR or EPMA, the 1000s of portable devices that need to talk to it can do so. This in turn requires that the hospital has a policy to manage technology, and is standardising on devices that it requires to connect in to the central system.
Re-usable electronic medical devices typically last for between 5 – 15 years, with perhaps 8-10 years being most common. However technological developments may now be rendering devices obsolete due to a lack of connectivity. It is not only devices such as infusion pumps and patient monitors that need to connect in to the Electronic Patient Record (EPR); most devices from weighing scales to diagnostic scanners are becoming available with wifi options. However, connection to the hospital network is not the only reason to think carefully about how devices are specified.
Hospital at home services
Once we turn our thoughts to hospital @ home services, we need to consider whether the devices that are used by trained professionals in our hospitals are suitable for use in a home setting. Official figures show that more than 8,000 drug errors cause serious harm or death in the UK each year, even when used by professional users. Once we move medical devices into the home environment, how does a non-professional patient or carer deal with problems or unexpected issues? ANSI/AAMI HE 75 defines the process for human factors engineering process for medical devices as starting with “define intended use, users and environment”. Up and down the country however, the urgency to create more capacity is leading to patients being sent home with inappropriate devices that were never designed for use in the home.
Planning Investment in healthcare technology
Guidance from the MHRA in England states that all hospitals should have a Board Member with responsibility for medical devices, and that there should be an organisation-wide medical devices group at which policy and investment decisions are made.(5) The guidance stops short of saying that the group should report directly to the hospital board, however with the level of reliance now placed on healthcare technology, it is hard to see how responsible organisations would fail to ensure that there is a direct reporting route. For clarity, this group should have membership that understands the strategic objectives of the organisation, and should be charged with determining the long term investment in medical technology to deliver those objectives. This requires that there is good understanding of current technological capabilities, and of emerging technologies and developments.
The clinical engineering service in large hospitals should be where the experts who can fulfil this role are found. Duality of provision means that some hospitals will choose to outsource technology provision to private contractors, and whilst there are many technology companies who can provide a wide array of technology based services, hospitals need to have in-house expertise to guide and act as internal “intelligent customers”. Technology management is no longer (if it ever was) a case of fixing broken items and conducting regular safety tests. Clinical engineers have the knowledge base and skill set to help hospitals make the best use of resources by specifying and planning for technology that can transform healthcare. Technology development is not a fad that is going to go away; the landscape has changed, and hospitals need to recognise and develop clinical engineers who can help them make the best use of the huge investment in technology that is required.
Artificial Intelligence
Going one step further, after acquisition of diagnostic information from the patient, this data can be processed by algorithms in so-called Artificial Intelligence (AI) software to provide both verification of clinical interpretation, but now also to recommend courses of action. In cases where rapid treatment is vital to outcomes but access to experienced experts is not always immediately available, e.g. stroke patients, there are now AI systems that will recommend a course of action to more junior staff on how to take prompt action.
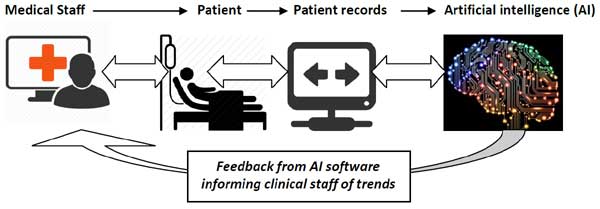
In a world of limited resources, the use of AI systems can provide diagnostic support that helps to facilitate prompt decisions when scarce medical staff are not available or are with other patients that really need them.
Fundamental questions of patient safety
Healthcare organisations face fundamental questions of patient safety, budgetary limits, and the ever-increasing need for availability of medical technology. Through the application of technology we can apply new ways of working where nurses and doctors can be supported by artificial intelligence to do their work thus improving patient safety. This is not science fiction, the AI systems are already available, but connectivity and an aging fleet of medical equipment does not allow this latest technology to be utilised. Enabling the best technology to be utilised in the best way needs to be guided by policy to ensure investment planning delivers productivity and patient care targets.
Healthcare technology management and policy is a serious issue recognised by research undertaken worldwide, including research carried out by the World Health Organisation.(6) There is a lack expertise in Healthcare Technology Management, and this is pointed out by many different organisations such as; the National Audit Office, the National Patient Safety Agency, the Medicines and Healthcare Products Regulatory Agency and the World Health Organisation.
Healthcare technology transformation is an extremely complex area within the healthcare environment covering the procurement, training, maintenance and governance of medical devices (including software systems) ranging from magnetic resonance imagers (MRI) to a complex AI software tools.
Clinical engineers are needed to enable the organisation to deliver sustainable transformational technology projects.
Bibliography
1. Nuffield Trust. Delivering the benefits of digital health care. [Online] 2016. [Cited: March 25, 2016.]
(View here.)
2. Broomfield, Matt. Budget 2016: George Osborne cuts £1.1bn from NHS repairs fund. [Online] 2016. [Cited: March 26/3/16, 2016.]
3. HM Government. Personalised Health and Care 2020. London : HM Government, 2014.
4. Carter, Lord. Operational performance and productivity in English HNS Acute Hospitals. London : Department of Health, 2016.
5. Medicines & Healthcare products Regulatory Agency. Managing Medical Devices - Guidance for healthcare and social service organisations. London : MHRA, 2015.
6. Petkova, Hristina. Barriers to innovation in the field of medical devices. Netherlands : World Health Organisation, 2010.