The past:
- In the 1950's there were no Electronic and Biomedical engineering departments (EBME) in the National Health Service (NHS).
- In the 1960's, the Department of Health and Social Security published Hospital Technical Memorandum number 8 called "safety code for electro-medical apparatus". The purpose of the document was to establish adequate standards for the design and construction of electro-medical apparatus since no other relevant national standard existed at the time.
- By the 70's some NHS hospitals had started thinking about maintenance and management of medical equipment. The NHS produced guidance in 1977 that was not implemented in the NHS but did raise the profile of medical engineering. In 1979, HTM 8 was superseded by the British Standard BS 5724 part 1. This document is a comprehensive specification for safety of medical electrical equipment.
- In the 80's the DHSS issued HEI 95 entitled "Code of practice for acceptance testing of medical electrical equipment". BS 5724 part 1 was also revised, making it identical to the International Electro-technical Commission standard IEC 601-1: 1988. The standard was subsequently re-numbered as IEC 60601-1.
- In the 90'sThe HEI 95 document was officially withdrawn on the publication by the Medical Devices Agency of MDA DB9801 - Medical device and equipment management for hospital and community based organisations. This document described a more strategic management approach to delivering EBME services.
- In November 2006, the MHRA published Device Bulletin DB2006(05).
- In 2010, the Care Quality Commission issued Regulation 16, outcome 11. Safety and suitability of equipment.
- In 2011, The World Health organisation issued guidance on Health Technology.
- In 2015, the MHRA issued ‘Managing Medical Devices - Guidance for healthcare and social services organisations’ (April 2015) https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/421028/Managing_medical_devices_-_Apr_2015.pdf
- In 2018, The National Health Service Litigation Authority became ‘NHS Resolution’. https://resolution.nhs.uk/
MHRA DB 2015 : Managing Medical Devices - Guidance for healthcare and social services organisations
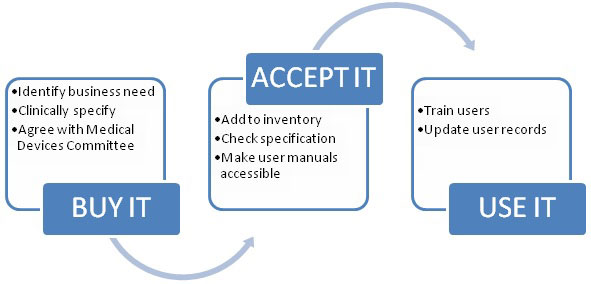
The purpose of this document is to outline a systematic approach to the acquisition, deployment, maintenance (preventive maintenance and performance assurance), repair and disposal of medical devices.
This Device Bulletin replaced previous guidelines published in:
- DB2006(05) – ‘Managing Medical Devices - Guidance for healthcare and social services organisations’
- DB 9801 ‘Medical device and equipment management for hospital and community-based organisations’ (including supplement 1 'Checks and tests for newly-delivered medical devices' and supplement 2 'Guidance on the sale, transfer of ownership and disposal of used medical devices')
- DB 2000(02) ‘Medical devices and equipment management: repair and maintenance provision’.
- DB 2003(05) 'Management of medical devices prior to repair, service or investigation'
- DB 2003(06) 'Community equipment loan stores - guidance on decontamination'
It was intended primarily for people in hospital and community based organisations (including social services) that are responsible for the management of medical devices, to help them set up systems that minimise risks associated with the use of those medical devices.
The purpose of this document is to outline a systematic approach to the purchasing, deployment, maintenance, repair and disposal of medical devices.
This guidance aims to:
- help healthcare organisations ensure the provision of adequate quantities of medical devices that are competently used by trained users, properly maintained and effectively managed to provide quality healthcare. The principles are summarised in Outcome 11 of the Care Quality Commission standards
- help healthcare organisations meet the Care Quality Commission’s standards (or equivalent) on prevention of incidents by unsafe or unsuitable devices
- help healthcare organisations to plan and co-ordinate sustainable replacement programmes
- help healthcare organisations optimise cost, risk and performance of medical devices
- address strategies for ownership and use of medical devices
- provide balanced information to help groups developing local policy
- identify relevant legislation
- identify sources of additional guidance
The main topics cover the life-cycle management of medical devices:
- management of medical devices
- acquiring appropriate devices
- training
- maintenance and repair
- reporting adverse incidents
- decontamination
- decommissioning and disposal.
It also states
"Responsible organisations should appoint a director or board member with overall responsibility for medical device management. There should be clear lines of accountability throughout the organisation leading to the board. These lines of accountability should be extended, where appropriate, to include general practitioners, residential and care homes, community based services independent hospitals providing services for NHS patients, managed care providers, PFI organisations and other independent contractors. It is important to establish who is accountable, and where there is a need for joint accountability arrangements".
The board should ensure that policies address:
- decontamination
- equipment life cycle
- procurement
- records
- adverse incident reporting
- actions required on MHRA's Medical Device Alerts and manufacturers' corrective notices
- training
- technical specifications
- regulatory compliance and related issues
- rationalisation to single models, where possible
- risk management
- equipment inventory
- manufacturer's instructions
In the past few decades some NHS Hospitals have been involved in producing these standards, and are therefore up to date, and others have been given documentary advice through the NHS executive, National Audit Office [NAO], Medicines and Healthcare Products Regulatory Agency [MHRA] (previously the Medical Devices Agency - MDA), and asked to implement the recommendations without management training or external assistance.
Many NHS organisations have not funded the increased responsibilities thereby leaving their EBME services struggling to keep up with the latest NHS executive and Dept of Health initiatives.
This has led to some EBME services still operating with the same number of staff that they had in the 1980's, but with a much larger inventory of medical equipment to manage and maintain, and increased expectations from top down. Unfortunately, many of the top executives have little or no understanding of the complexity of meeting the standards
The National Audit Office produced a report on The Management of Medical Equipment in NHS Acute Trusts in England.
See: http://www.nao.org.uk/publications/nao_reports/9899475.pdf
The report concludes that, although there are examples of good practice, overall more needs to be done by trusts to allocate clear responsibility for medical equipment at board level. Trusts need to ensure that inventory information is comprehensive and used fully in decision making. Procurement of medical equipment needs to be better co-ordinated across trusts, with more involvement of technical personnel. They can also usefully contribute to non-clinical aspects of user training. We also recommend action that should help to improve the standards of reporting of adverse safety incidents, and to reduce their occurrence.
The risks of a Hospital not investing in its EBME team are:
- Patients may die
- Longer bed stays / bed management problems
- Higher costs for capital purchase
- Higher costs for revenue purchases
- Higher costs for externally contracted maintenance
- Higher legal fees
- Higher NHSLA (CNST) costs
The actual cost of doing nothing... i.e. not investing in EBME services is higher than the investment need to have a service that meets the needs of the 21st century.
The future for EBME managers:
EBME managers will hold a senior management post, but will be expected to:
- Keep up with the latest innovations
- Advise on all aspects of medical devices management in line with National Audit office and MHRA recommendations.
- Understand clinical interactions between device users, and patients.
- Manage larger multi disciplinary teams
- Advise Trust executives / attend boardroom meetings.
- Liaise with other senior managers in procurement, governance, finance, infection control, etc.
An EBME manager in the 21st century is expected to be able understand and deliver:
- Planned equipment replacement programmes
- Tender advice to head of procurement
- MHRA liaison (Incident investigation)
- Advising medical and nursing directors on serious untoward incidents (SUI's)
- NHSLA advice for medical devices governance
- Policy production
- Evaluating devices
- https://www.ebme.co.uk/articles/management/ebme-medical-device-evaluation
- Commissioning devices
- Planned maintenance
- Remedial Maintenance
- User training
- Technical training
- Database reporting and management
- Income generation through service level agreements with other Hospitals.
- Service contract assessment and control
- Cost saving improvements
- Equipment library set up and management
- Gas safety
- Electrical safety
- Technician training
- Disposal
- Quality management systems
It is a challenge for the EBME manager to keep up with their continuing professional development. It is important for senior executives to understand what the risks are of not supporting the EBME manager (in delivering professional services).
The future innovations in medical device management should be coming from proactive EBME managers, with executive support.
World Health Organisation (WHO)
A paper entitled ‘Development of medical devices policies’ was published by the World Health Organisation that is specifically relates to the development of medical devices policy at country level. This 2011 paper discusses the importance of medical devices, recognising the important role of health technologies in the prevention, diagnosis, and treatment of illness and disease, as well as patient rehabilitation. This document talks about policies, strategies and action plans. This WHO paper is aimed at national / government level to raise the awareness that every country should have a medical technology policy. The World Health Organisation goes on to say in their conclusion:
Data shows that policies, strategies, and action plans for medical devices are being developed in member states. …. Recommendations from the first global Forum on medical devices in 2010 will continue to raise the awareness of the crucial role that medical devices play in the prevention, diagnosis, and treatment of disease and rehabilitation. The hope is that the higher profile of medical devices will translate into better healthcare for the global population, allowing them to enjoy a better quality of life.
Poor procurement leads to variation and ultimately higher risk to the patient. This is recognised by research carried out by the World Health Organisation:
‘A number of studies have shown that between 39% and 46% of adverse events resulting from misuse of medical devices take place in the operating room. In most of these studies, the cause is only indicated as operation related. …Variation in medical devices between hospitals (and even within the same hospital) is one of the causes of these accidents.’
The World Health Organisation recognises the benefits of medical equipment in their paper ‘Increasing complexity of medical technology and consequences for training’, but also point out the risks of underestimating the importance of training:
‘The influence of the operator on the effective and safe application of medical technology is generally underestimated. In an investigation on incidents involving defibrillators in the US, it was concluded that the majority of the incidents were due to incorrect operation and maintenance. A study of 2000 adverse incidents in operating theatres in Australia showed that only 9% were due to pure equipment failure. In two reports on the use of critical care equipment by nursing staff, 19% and 12.3% of nurses, respectively, indicated that they had used equipment improperly, which had consequently harmed a patient’.
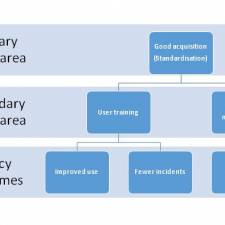
Good management policy for acquisition, training, and maintenance can results in better use and utilisation of the assets, reducing the risks to patients, and improving outcomes for patients. The acquisition and use of medical technology has a major impact on the business of the trust and the outcomes for patients.
I would recommend all EBME Managers attend the annual EBME conference where they will meet like minded colleagues.
See: https://www.ebme.co.uk/seminar
Compiled and Edited by Dr John Sandham CEng MIET FIHEEM
Sources:
http://www.mhra.gov.uk/
http://www.cqc.org.uk/
http://www.who.int/medical_devices/management_use/en/
http://www.nao.org.uk/
https://resolution.nhs.uk/